Download Pdf (Link given below)
PHYSICAL CHANGE & CHEMICAL CHANGE
| Basis | Physical Change | Chemical Change |
| New substance | Not formed | Formed |
| Type of change | Physical properties change | Chemical composition changes |
| State/appearance | Size, shape or state changes | Entire nature of substance changes |
| Reversibility | reversible | irreversible |
| Chemical reaction | Not a chemical reaction | It is a chemical reaction |
| Energy change | Very less energy change | Energy absorbed or released |
| Chemical bonds | No bond breaking or formation | Bonds break and new bonds form |
| Mass | Mass remains same | Mass conserved (reactants = products) |
| Effect on properties | No new properties | New properties appear |
| Example | Melting of ice | Rusting of iron |
CHEMICAL REACTION
Process in which new substance are formed with new chemical (may be physical also), is/are formed, is called chemical reaction.
*CHARACTERISTISTICS OF CHEMICAL REACTION
1. Evolution of Gas
- Zinc reacts with dilute hydrochloric acid to produce hydrogen gas.
Zn + 2HCl → ZnCl₂ + H₂↑ - Calcium carbonate reacts with dilute hydrochloric acid to release carbon dioxide gas.
CaCO₃ + 2HCl → CaCl₂ + H₂O + CO₂↑
2. Formation of Precipitate
- Lead nitrate solution reacts with potassium iodide solution to form a yellow precipitate of lead iodide.
Pb(NO₃)₂ + 2KI → PbI₂↓ + 2KNO₃ - Silver nitrate reacts with sodium chloride to form a white precipitate of silver chloride.
AgNO₃ + NaCl → AgCl↓ + NaNO₃
3. Change in Temperature
- Quick lime reacts with water and releases heat (exothermic reaction).
CaO + H₂O → Ca(OH)₂ + Heat - Burning of magnesium ribbon produces heat and light.
2Mg + O₂ → 2MgO + Heat
4. Change in State
- Hydrogen gas reacts with oxygen gas to form liquid water.
2H₂ + O₂ → 2H₂O - Carbon dioxide gas reacts with aqueous calcium hydroxide to form solid calcium carbonate.
Ca(OH)₂ + CO₂ → CaCO₃↓ + H₂O
5. Change in Colour
- Iron displaces copper from copper sulphate solution, changing the colour from blue to green.
Fe + CuSO₄ → FeSO₄ + Cu - When copper is heated in air, it turns black due to formation of copper oxide.
2Cu + O₂ → 2CuO
DOWNLOAD PDF of this Short Notes from telegram channel – CLICK HERE
LAW OF CONSERVATION OF MASS
– Given by Lavoisier in 1774
– It states that:
- Mass is neither be created nor be destroyed.
- Total mass of Reactant = Total mass of Product
MAKING EQUATIONMORE INFORMATIVE
1. Indicating “Physical State”
2. Indicating heat changes (+heat)
3. Indicating the conditions
TYPES OF CHEMICAL REACTION
1. Combination Reaction (A + B = AB)
- Calcium oxide reacts with water to form calcium hydroxide.
CaO + H₂O → Ca(OH)₂ - Magnesium burns in air to form magnesium oxide.
2Mg + O₂ → 2MgO
2. Decomposition Reaction (AB = A + B)
- Calcium carbonate decomposes on heating to form calcium oxide and carbon dioxide.
CaCO₃ → CaO + CO₂ - Silver chloride decomposes in sunlight to form silver and chlorine gas.
2AgCl → 2Ag + Cl₂
#Types of Decomposition Reaction
1)Thermal Decomposition
2)Electro Decomposition
3)Photo Decomposition
3. Displacement Reaction (A + BC = AC + B)
More reactive displaces less reactive
#Reactivity Series:
It is a series in which metals arranged in decreasing order of their reactivities on moving from top to bottom.
Trick to Learn: Kaka Nana Chaat Mange Aloo Zara Feke Pade Hai CHAAP.
METAL NAME (Reactivity decreases as we move down)
Kaka – K (Potassium)
Nana – Na (Sodium)
Chaat – Ca (calcium)
Mange – Mg (Magnesium)
Aloo – Al (Aluminium)
Zara – Zn (Zinc)
Feke – Fe (Iron)
Pade – Pb (lead)
Hai – H (Hydrogen)
C – Cu (Copper)
H – Hg (Mercury)
A – Ag (Silver)
A – Au (Gold)
P – Pt (Platinium)
Example:
- Zinc displaces copper from copper sulphate solution.
Zn + CuSO₄ → ZnSO₄ + Cu - Iron displaces copper from copper sulphate solution.
Fe + CuSO₄ → FeSO₄ + Cu
4. Double Displacement Reaction (AB + CD = AC +BD)
A reaction in which exchange of ions takes place between two compounds.
- Lead nitrate reacts with potassium iodide to form lead iodide and potassium nitrate.
Pb(NO₃)₂ + 2KI → PbI₂ + 2KNO₃
- Sodium sulphate reacts with barium chloride to form barium sulphate and sodium chloride.
Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
5. Redox Reaction
A reaction in which oxidation and reduction take place simultaneously.
*Reduction: Adding H₂ OR Removing O₂ OR Both
*Oxidation: Adding O₂ OR Removing H₂ OR Both
- Zinc reacts with copper sulphate solution.
(Zinc is oxidised and copper ions are reduced)
Zn + CuSO₄ → ZnSO₄ + Cu
- Copper oxide reacts with hydrogen to form copper and water.
(Copper oxide is reduced and hydrogen is oxidised)
CuO + H₂ → Cu + H₂O
DOWNLOAD PDF of this Short Notes from telegram channel – CLICK HERE
EFFECTS OF OXIDATION ON EVERYDAY LIFE
1) CORROSION:
Process of slowing eating up of metals due to their conversion into oxides, carbonates, etc by the action of air.
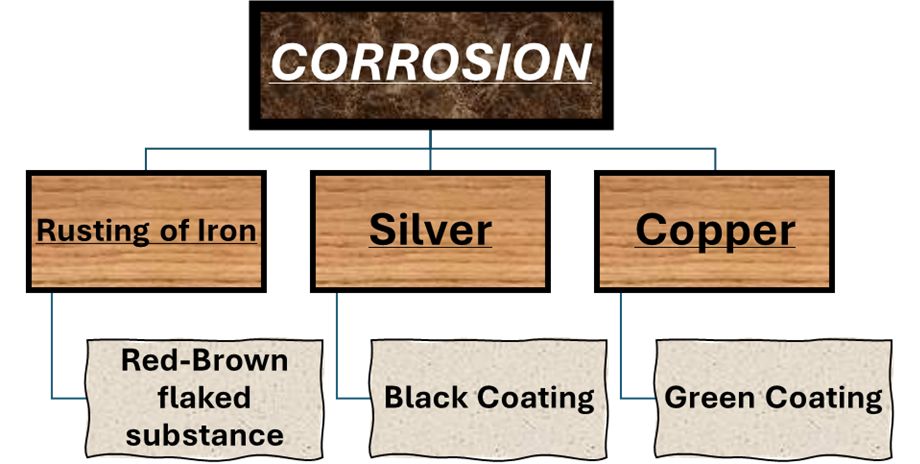
*Prevention from Corrosion:
- Painting
- Oiling
- Galvanisation (Zinc on Iron)
2) RANCIDITY:
Fats and Oil present in food get oxidized.
*Prevention:
- Adding Anti-Oxidants: BHA and BHT
- Packaging fat and oil containing food in Nitrogen gas
- Keep food in air tight
- Keep food in cool places
- Keep food in dark places
- Vacuum packing
IMPORTANT EXPERIMENTS
1) BURNING OF MAGNESIUM
- First Clean – To remove magnesium oxide layer.
- Burn with dazzling white flame
- White ash formed (magnesium oxide)
- 2Mg + O₂ = 2MgO
2) CASE OF WHITE WASHING
Substance – Calcium Oxide (CaO)
* Formation of Slaked Lime
When quicklime reacts with water, slaked lime is formed:
CaO + H₂O → Ca(OH)₂
This reaction is exothermic (heat is released).
* Reaction during Whitewashing
When slaked lime is applied on walls, it reacts slowly with carbon dioxide from the air to form calcium carbonate:
Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
3) TEST FOR H₂ GAS
- Bubbles of H₂ gas.
- Burn with pop sound.
4) IRON NAILS IN COPPER SULPHATE SOLUTION
- Blue solution turn green
- Brown Copper deposited
- Fe + CuSO₄ → FeSO₄ + Cu
5) RUSTING IS FASTER IN COASTAL AREAS
- Presence of Moisture and salt
- Increases Corrosion rate
DOWNLOAD PDF of this Short Notes from telegram channel – CLICK HERE
Explore other chapter’s NOTES – CLICK HERE or visit Gyanora.in
For Practicing chapter wise question answers – CLICK HERE or visit QnA.Gyanora.in
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6
